The incomplete absorption can be due to some of the incident light being transmitted through the body or to some of it being reflected at the body’s surface. Since the absorptivity is less than unity for real objects, a real object can not absorb all incident light. That is, a blackbody is a perfect absorber. Like emissivity, value of absorptivity is in the range 0 < α < 1.įrom its definition, a blackbody, which is an idealized physical body, absorbs all incident electromagnetic radiation, regardless of frequency or angle of incidence. These collectors waste very little solar energy through the emission of thermal radiation.Īnother important radiation property of a surface is its absorptivity, α, which is the fraction of the radiation energy incident on a surface that is absorbed by the surface. For example, solar heat collectors incorporate selective surfaces with very low emissivities. Emissivity plays an important role in heat transfer problems. Real objects with emissivities less than 1.0 (e.g., copper wire) emit radiation at correspondingly lower rates (e.g., 448 x 0.03 = 13.4 W/m 2). The surface of a blackbody emits thermal radiation at the rate of approximately 448 watts per square meter at room temperature (25 ☌, 298.15 K). Emissivity is simply a factor by which we multiply the black body heat transfer to consider that the black body is the ideal case. 
Quantitatively, emissivity is the ratio of the thermal radiation from a surface to the radiation from an ideal black surface at the same temperature as given by the Stefan–Boltzmann law. The Stefan-Boltzmann law must include emissivity to consider that real objects are gray bodies. Real objects do not radiate as much heat as a perfect black body, and they radiate less heat than a black body and therefore are called gray bodies. The emissivity, ε, of the surface of a material is its effectiveness in emitting energy as thermal radiation and varies between 0.0 and 1.0.īy definition, a blackbody in thermal equilibrium has an emissivity of ε = 1.0. For example, solar radiation reaches the surface of the earth after passing through cold layers of the atmosphere at high altitudes. In contrast to heat transfer by conduction or convection, which occurs in the direction of decreasing temperature, thermal radiation heat transfer can occur between two bodies separated by a medium colder than both bodies. 
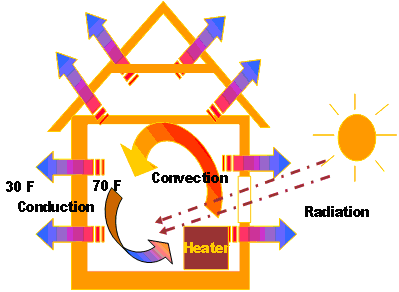
Energy transfer by radiation is fastest (at the speed of light), and it suffers no attenuation in a vacuum. Thermal radiation does not require any medium for energy transfer. It is generated by the thermal motion of charged particles in matter, and therefore any material with a temperature above absolute zero gives off some radiant energy. The term thermal radiation is frequently used to distinguish this form of electromagnetic radiation from other forms, such as radio waves, x-rays, or gamma rays. Thermal radiation is electromagnetic radiation in the infra-red region of the electromagnetic spectrum, although some of it is in the visible region. It can be expressed by the following equation: Radiation heat transfer rate, q, from a body (e.g., a black body) to its surroundings is proportional to the fourth power of the absolute temperature. Thermal radiation is electromagnetic radiation in the infra-red region of the electromagnetic spectrum, although some are in the visible region.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
